|
When Nature ferments grapes, or any other fruit for that matter, wine is
not the end product. Instead, unpleasant concoctions containing
vinegars, mercaptans and other substances are formed, with the final end
being water and assorted solids and gases. Although most good winemaking
involves interfering with Nature as little as possible nonetheless we
need to steer her a bit, and in fact completely stop some natural
processes at just the right moment.
An indispensable ally of the winemaker in achieving these things is
sulphur dioxide. We will refer to it by its
chemical formula,
SO2. In this article we will be investigating
how to use
SO2 to do the following things for us: inhibit wild and spoilage
yeasts and unwanted bacteria (this can include the malolactic bacteria
at sufficiently high
SO2 levels); help prevent oxidation and preserve fruity flavour and
freshness in wine.
Sourcing SO2
SO2
is a pungent,
choking gas which is somewhat soluble. The most practical source for the
home winemaker is the salt, potassium metabisulphite,
which is 57% SO2
Since you can detect SO2
when you smell a sample of potassium metabisulphite it is
evident that the solid decomposes easily. This happens on contact with
carbon dioxide and moisture in the air. Keep your container of potassium
metabisulphite tightly closed to minimize this problem. In any case, you
probably shouldn’t keep the stuff around for more than a year before
buying fresh.
“Campden” tablets are made of potassium metabisulphite. Each
tablet, when fresh, contains 0.44 grams of it. However, if they are old,
a lot of the SO2
will have been lost and their effects will be unreliable. You’re better
off to use bulk potassium metabisulphite. It’s cheaper too.
Sodium metabisulphite is also a source of
SO2 but probably
should be used only for equipment sterilizing purposes,
not in must or wine. For one thing, many people avoid sodium in their
diets, for another, the presence of potassium ions in wine is more
useful than sodium.
Sometimes an
old fashioned winery will burn a sulphur stick in empty
barrels to keep them sterile. Under no circumstances should the home
winemaker ever do this. The presence of any elemental sulphur, such as
might drip into the barrel will lead inevitably to the formation of the dreaded hydrogen
sulphide. In the winemaking business,
-ite sulphur compounds are friendly,
-ides are deadly enemies.
Under a very few circumstances, solid potassium metabisulphite
may be used directly. For instance if you decide to add
SO2 to red grapes
before crushing, a scant ¼ teaspoon sprinkled on a 36 pound lug of
grapes will give you about what you need—somewhere around 30 to 40 parts
per million SO2.
Don’t do this with white grapes or when using red grapes to
make a rosé. When you press, the
SO2 will wash off into the juice in uncontrollable
amounts and you will likely have far too much in the free run, and next
to none in the pressed portion.
The 10% Solution
A much better way to get your
SO2 is from a 10% solution of potassium metabisulphite in
water. For instance, you could add water to 1 pound of potassium
metabisulphite to make a total volume of 1 imperial gallon. Or, if you
prefer metric, add enough water to 100 grams of potassium metabisulphite
to make up a total volume of 1.00 litres. Fresh 10% solution is 5.7%
SO2.
A commonly used unit of measurement for
SO2 in must or wine is “parts per million” or “ppm”. 1
ppm is the same as 1 milligram per litre. I will use ppm.
For example, if you add 2.4
millilitres
of 10% potassium metabisulphite solution to 1.0 imperial gallons of wine
you will be adding 30 ppm SO2.
If you have a 19.2 litre carboy to which
you wish to add 20 ppm SO2,
multiply 0.35 by 19.2 to get an
SO2 addition of 6.7 mL of 10% solution. Consider making
up your own spreadsheet giving SO2
additions for your own sizes of barrels and carboys.
Putting SO2 to good use
You might hear a commercial winemaker tell you that she
“doesn’t use any SO2
at all until after the primary ferment is complete, particularly
with white wines.” Such a winemaker knows the complete history of her
grapes—exactly where they came from and how they were handled. She
undoubtedly also has elaborate handling equipment—must coolers, inert
gas covered tanks and all the rest. You should know a lot about what
you’re doing before you decide to postpone adding
SO2 until some
middle point in the winemaking process.
Let’s start with red grapes. You need to suppress any bacteria
and wild yeasts they may have picked up, prior to inoculating with a
selected yeast culture. If you try to depend on wild yeasts, they will
likely die before all the sugar is fermented out, leaving you with a
sticky problem or worse. Vinegar bacteria can produce an undesirable
amount of ethyl acetate in the early part of the fermentation if not
checked.
You probably bought the grapes by the pound and can assume
around 5 litres of finished wine from each 20 pounds. Addition of 2.7
millilitres of 10% potassium metabisulphite solution for each 20 pounds
works out to 30 ppm SO2.
If the grapes are in reasonable shape, this should do the job for you.
If your grapes are in perfect shape and the pH is low enough, you can do
with less. We will deal with pH considerations later.
If you are planning to have a malolactic ferment, or ML,
happen at the same time as the sugar ferment, don’t add the ML culture
until the sugar ferment is well underway. By that time enough of the
SO2 will have gone so that the ML bacteria can multiply
and flourish. Alternatively keep your
SO2 addition down
to, say, 20 ppm. We’ll talk more about ML when we discuss white wines.
If you are concerned about excessive mould, possibly accompanied by traces
of vinegary smells, increase the
SO2 addition to 50 or 60 ppm or in extreme cases even
more.
The
SO2 you add will
also lead to production of small quantities of glycerol in the early
part of the ferment. This is generally desirable.
When you make white or rosé wine the situation is a bit
different. Grape skins contain phenols. These add flavour and colour to
wine. They can also contribute astringency, bitterness and browning.
These things are of more concern in whites and rosés than in red wines.
SO2
can contribute to phenol extraction from the skins and this is
another reason it shouldn’t
be added to a white or rosé
until after the pressing has been done. However, the addition should be
made promptly since white must quality suffers from oxygen absorption
from the air. As soon as you have pressed, you have an accurate measure
of your yield and can thus calculate the
SO2 addition more
precisely.
How much
SO2?
How much to add depends on a number of factors. What was the
condition of the grapes? What is the pH? (We shall see later, how
SO2 is more
effective at lower pH). Are you planning on putting the wine through a
malolactic ferment? Is the juice intended for making a champagne method
sparkling wine?
30 ppm SO2
for juice from sound fruit with a pH of 3.4 or so, and destined for
a regular wine should be fine.
If you hope to have a malolactic ferment happen along with the
sugar ferment, you likely have a higher acid Chardonnay, or something,
say around pH 3.2. Smaller SO2
additions are okay here—say 20 ppm. Malolactic bacteria won’t
work at levels higher than around 15 ppm, but by the time you add an ML
culture, much of the SO2
will have been used up.
Juice destined for Champagne method wine will probably have a
low pH, close to 3.0, say. You are going to want to have a malolactic
ferment occur. If the grapes were perfect, you might get away with no
SO2 at all until
the first racking. This however is a bit nerve-wracking, like having a
tooth filled without anaesthetic. The danger of some undesirable
oxidation of the must is there, so better to go with 10 ppm
SO2 or so.
At the other end of the scale, juice from grapes with a lot of
mould, possibly with some vinegary smells, should have 50 to 60 ppm
SO2 or even more
added. Who knows—maybe you have lucked on to some botrytised Riesling or
Semillon and plan a serious dessert wine. Botrytised grapes may require
100 ppm SO2 or
even more for adequate protection.
How about frozen or sterile packaged musts? With white or rosé
juice, you can either trust the shipper to tell you how much
SO2 was added, or
you can test and make additions accordingly.
It is difficult to test reds for SO2, because the red colouring matter interferes with the
chemical reaction involved in the test and also makes it difficult to
see the colour change involved. You pretty well have to trust the information on the shipping
label. The fact that testing reds for free
SO2 is difficult makes it imperative that you keep an
accurate record of all SO2
additions in order to be able to estimate the situation at any
given time.
The next time you are going to consider adding
SO2 to the wine is
at the first racking. In most cases, this will be after the sugar
fermentation is complete and the new wine is dry.
If you want to stop active fermentation to retain residual
sugar, don’t try to use SO2
as your main tool. A vigorous ferment of a strong yeast will
laugh at you and carry right on to the end. Selected combinations of
racking, fining, chilling and filtering are the way to go.
SO2 will be
involved, but only as it would be normally used in conjunction with
these other processes.
Stifling
Oxidation
An important reason for adding
SO2 when you rack is to avoid oxidation. It does this in
three main ways.
-
When you smell a wine that is oxidized, the chemical you are
smelling is acetaldehyde. SO2
combines with acetaldehyde to form a stable compound.
When there is oxygen around,
SO2 itself becomes oxidized before phenol compounds in
the wine do, and so acts as an oxygen scavenger.
SO2
suppresses the activity of enzymes that cause browning and other
problems.
|
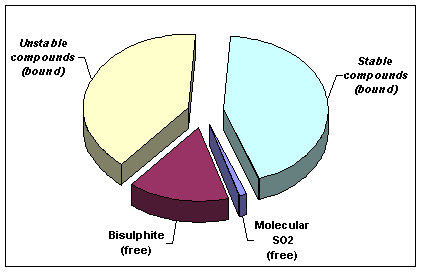
-
Chart showing typical relative amounts of free
and bound
SO2
|
So, when you add SO2
it doesn’t all hang around. Lots of it gets used up doing
these various jobs for you and becomes
“bound”.
The remainder remains “free”.
The bound portion consists of two parts. One part is made up of
irrevocably bound compounds with aldehydes and proteins. The other part
is made up of less stable compounds. These can partly turn back to the
free form when the existing amount of free is lowered, or even if
temperature is increased. This free portion also consists of two parts:
one is relatively inactive bisulphite and the other, smallest of all the
segments, as shown in the accompanying chart is
molecular This is
the crucial active portion and its size depends both on
pH and the total amount of
free SO2.
It is worth noting at this point that in the early stages of a wine,
when the total SO2
additions are less than 50 ppm or so, roughly half of further
additions remains free and half immediately becomes bound. Later, when
total additions are above about 60 ppm, most of any further addition
remains as free. This knowledge gives us further reason to keep good
records of SO2
additions, particularly in the case of reds, where direct measurement of
free SO2 is not
reliable.
Testing
for Free SO2
The test procedure that follows works well only for white or rosé wines.
Some of the colouring matter in red wines reacts to the test chemicals
in the same way as SO2
making the results pretty well meaningless.
It should be noted that SO2
testing kits may be available at your local winemaking supply
shop. Since they will contain all the necessary ingredients,
instructions and measuring vessels, you
will save yourself substantial effort by buying one. What follows
assumes you wish to put together your own kit.
You will need the following chemicals, which you might need some help
with. The chemistry teacher at your local high school might be receptive
to a contribution to his or her science department’s petty cash fund.
0.02 molar iodine solution: Accurately weigh out 2.54 grams of iodine. Roughly weigh 5
grams of potassium iodide. Add a few millilitres of distilled water,
barely enough to cover the chemicals, and agitate until the iodine is
completely dissolved. This may take a bit of time. Finally, add enough
distilled water to make an accurately measured 1.00 litres of solution.
Dilute sulphuric acid: Add about 250 millilitres of concentrated sulphuric acid to
about 750 millilitres of water.
Unless you have previous experience handling sulphuric acid, don’t even
think of doing this dilution yourself.
Starch solution: Add about 1 gram of starch to about 100 millilitres of water. Stir and
bring to a boil then cool.
To do the test: first fill a clean dry 6 or 10 mL syringe with the
iodine solution. Next, accurately measure out 50 mL of the wine to be
tested. Add 1 mL or so of starch solution and about 10 mL of dilute
sulphuric acid.
Immediately start adding iodine solution to the sample, swirling it as
you go. You will note a purple-black patch which disappears as you
swirl. As soon as the purple colour persists, stop adding iodine, and
note how many mL you’ve used. Multiply this by 12.8 to give you the
number of ppm of free SO2
in the wine.
Testing
for Total SO2
You will need some 10%
sodium hydroxide solution in addition to the chemicals required for the
free SO2 test. To make this
up, add enough water to 10 g of solid sodium hydroxide to bring the
volume up to 100 mL. Great accuracy here isn’t necessary. Mix
thoroughly.
To do the test: Accurately measure 20 mL of wine
and put it in a narrow necked container such as an erlenmeyer flask. Add
roughly 25 mL of 10% sodium hydroxide solution. Immediately cover the
container and allow to sit for 15 minutes. Fill a clean dry 6 or 10 mL syringe with 0.02 molar iodine solution. At the end of the
15 minutes, add 10 mL dilute sulphuric acid along with about 1 mL of starch solution to the sample and immediately
start adding iodine solution. Stop when the purple colour persists. Note
the volume of iodine solution used in mL, and multiply by 32. This is your total SO2
in ppm.
Stability of Chemicals.
The dilute sulphuric acid and 10% sodium hydroxide solutions are very
stable and will last for years. The sodium hydroxide should be stored in
high density plastic in preference to glass. The starch solution will
get mouldy. It should be replaced as soon as the slightest bit of
discolouration occurs. Iodine is highly volatile. The iodine solution
should be in as small a glass container as is convenient, and kept
tightly closed and in a cool place. An alternative method of
managing the iodine is to make up a 0.20 molar stock solution (10 times
working strength). From time to time make up as much working strength
(0.02 molar) solution as you will need for a month or so by diluting one
volume of the 0.20 molar stock solution with 9 times that volume of
distilled water.
Adding SO2 at racking
When racking red wines, depending on pH, the addition of from 20 to 30
ppm SO2
each time should do the trick nicely. For the first couple of rackings,
when the
total
SO2 added since the beginning is less than 50
ppm or so, about half of what you add immediately gets
bound, leaving half as free.
After your total additions
over the life of the wine add up to around
60 ppm or more, most of any
additional SO2
you add remains as free.
Be sure to pour the
SO2
solution into the bottom of the receiving container
first and then rack the
wine. This way the SO2
is around all the time to suck up unwanted oxygen.
If you have started a malolactic ferment as well and you are not certain
it has completed, you could go with less
SO2 at
racking—maybe 15 ppm, maybe only 10. In this case, your pH is likely to
be pretty low anyway and as we’re going to see later, that makes the
SO2 much more
effective.
You are, of course, keeping a good record of your
SO2 additions, aren't you? A reasonable rule of thumb for
red wines is to keep the total addition of
SO2 from crush to bottling at less than 150 ppm.
With white or rosé wines, test before racking, and add enough
SO2 to bring the free up to 20 or 30 ppm.
Once again, if there is a malolactic ferment involved and/or you are
going to do a bottle ferment later, for champagne method sparkling wine,
you want to keep the SO2
down. Since under these conditions, the pH is going to be low, you are
probably okay adding only 10 ppm or so.
A reminder about racking techniques is in order here. Always make sure
your syphon tube is down to the bottom of the receiving container. Don’t
splash the wine. If you trying to get away with minimal
SO2 and you have a carbon dioxide cylinder, purge the
receiving container of air with CO2 before adding
SO2 and
racking.
SO2 and pH
I have made several references to the connection between the
effectiveness of SO2
and pH. It is about time to explain how this works. 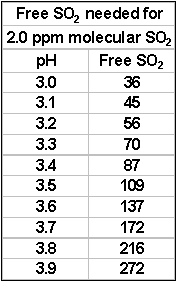
What is really protecting your wine is molecular
SO2.
 When you add SO2,
depending on circumstances, some of it immediately becomes bound.
What remains is called “free” and is in two parts. The larger, and
relatively ineffective free part is “bisulphite” (HSO3-). The smaller
part of the free is the active
molecular SO2.
The amount of molecular SO2
in your wine depends both on the level of free
SO2 present as
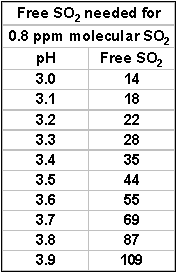
well as pH. For instance at pH 3.2 the amount of free
SO2 for 0.8 ppm molecular
SO2 is 22 ppm. At pH 3.5, you will need 43 ppm free –
essentially double. In most situations, 0.8 ppm molecular
SO2 during bulk storage and at bottling will provide you
with adequate protection from oxidation and bacterial action. This
includes prevention of ML bacteria as well—important if you’ve blended
ML affected wine with non-ML affected and require stability. It is
important to remember that the amount of free
SO2 in
the wine depends on three things: how much you added, how much was
present before the addition and how much of your addition promptly
becomes bound. In the case of whites and rosés, the best thing to do is
a free SO2 check.
In the case of reds, you need to do some good estimating, based on
previous SO2
additions as mentioned elsewhere in the article. When you add SO2,
depending on circumstances, some of it immediately becomes bound.
What remains is called “free” and is in two parts. The larger, and
relatively ineffective free part is “bisulphite” (HSO3-). The smaller
part of the free is the active
molecular SO2.
The amount of molecular SO2
in your wine depends both on the level of free
SO2 present as
well as pH. For instance at pH 3.2 the amount of free
SO2 for 0.8 ppm molecular
SO2 is 22 ppm. At pH 3.5, you will need 43 ppm free –
essentially double. In most situations, 0.8 ppm molecular
SO2 during bulk storage and at bottling will provide you
with adequate protection from oxidation and bacterial action. This
includes prevention of ML bacteria as well—important if you’ve blended
ML affected wine with non-ML affected and require stability. It is
important to remember that the amount of free
SO2 in
the wine depends on three things: how much you added, how much was
present before the addition and how much of your addition promptly
becomes bound. In the case of whites and rosés, the best thing to do is
a free SO2 check.
In the case of reds, you need to do some good estimating, based on
previous SO2
additions as mentioned elsewhere in the article.
The level at which molecular SO2
can be detected by the human senses is about 2.0 ppm. This is also
the level which is needed for maximum protection of your wine. This is
particularly true in the case of sweet, and most notably, botrytised
wines
Using Potassium Sorbate
Sometimes one wishes to finish a wine with some residual sugar
left—Riesling, Gewürztraminer, Muscat Canelli and Chenin Blanc are among
the grapes that lend themselves particularly well to this. In order to
prevent renewed fermentation
after ferment has been stopped,
00 to 250 ppm potassium sorbate is often used. The effectiveness of
potassium sorbate is pH dependent. To get close to the same
effectiveness from a given dose of potassium sorbate would require
around 55 ppm of free SO2
at pH 3.6 as opposed to only 28 ppm at pH 3.3.
It is essential when using sorbate to have effective
SO2 levels high enough to prevent a malolactic ferment
from happening. If ML occurs in the presence of sorbate, a peculiarly
revolting geranium-like smell is produced for which, alas, there is no
remedy. The wine is a goner.
Bottle Rinsing with SO2
I often find it useful to use an
SO2 bottle rinse when I am bottling. The rinse
solution is 50 mL of 10% potassium metabisulphite solution made up with
water, to about 750 mL in a winebottle. I have tested the effect of this
several ways, and consistently find that after rinsing, and draining the
bottles for about a minute, the free
SO2 added is close
to 8 ppm. Curiously, this is true for both 750 mL and 375 mL bottles.
This is a useful way of adding a touch of
SO2 at bottling
time, particularly if the carboy you’re bottling has a bit of sediment
and you don’t wish to stir it , or subject it to one more racking.
The use of SO2
started with the Romans, and I’m sure there isn’t a self respecting
winery in the world today that gets away without it
***
Potassium metabisulphite is K2S2O5.
For the sake of simplicity, we will consider that it breaks down
in acidic solution into two K+ particles and one S2O5=
particle. The two K+’s probably wind up attached to tartarate
particles and settle out as potassium bitartarate. They are of no
further concern to us here. The metabisulphite particle reacts with two
hydrogen particles – this is an acidic solution we’re working with – to
produce two sulphur dioxide molecules and one water molecule:
S2O5= + 2H+
® 2SO2 + H2O
If you care to
look up the atomic masses and do the arithmetic, you will see that
potassium metabisulphite is thus, about 57% SO2.
_________________________
When using a sulphite solution as a sterilizing agent, such as for
rinsing pumps or storing primary fermenters, be sure to add a little
tartaric acid in order to bring the pH down and increase the level of
molecular SO2.
_________________________
An excellent way to measure small quantities of liquids accurately is to
use a medical syringe. I have a set running from a 60 mL one all the way
down to a 1 mL size, calibrated in hundredths. The 1 mL syringe, needle
tipped, is fine enough to add a controlled amount of
SO2 to a single
375 mL half-bottle.
_________________________
[4]
SO2 + H2O Û HSO3- + H+ . An accepted equilibrium constant
in this equation is 1.77. The accompanying tables are based on
this equation.
_________________________
[5]
Neither SO2 nor sorbate will stop an active fermentation alone.
This can only be achieved with chilling, settling, racking and
filtration using appropriate SO2 additions as adjuncts to these
operations.
© Charles Plant 2001 |



